Comparative study of two steam reforming kinetic models for pure hydrogen production from ethanol in a separating reactor
Main Article Content
Abstract
The present work deal with a comparative study of two kinetics models of ethanol conversion by steam reforming for pure hydrogen generation in a Pd-Ag based membrane reactor. For this purpose, a mathematical model describing the reaction and separation phenomena that take place simultaneously in the membrane reactor was developed. The numerical solution of the set of differential equations constituting the obtained model allows us to quantify the process performance over both 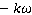 and
and  catalysts. According to the obtained results, it was found that the performance of the ethanol steam reforming process depends strongly on the catalyst nature. In addition, the process's strongly sensitive to the used operating conditions, especially hydrogen partial pressure in permeate and in reaction sides, in which a good correlation and a synergistic effect between these parameters could be established. Under the optimal operating conditions, the quantity of hydrogen recovered when using the
catalysts. According to the obtained results, it was found that the performance of the ethanol steam reforming process depends strongly on the catalyst nature. In addition, the process's strongly sensitive to the used operating conditions, especially hydrogen partial pressure in permeate and in reaction sides, in which a good correlation and a synergistic effect between these parameters could be established. Under the optimal operating conditions, the quantity of hydrogen recovered when using the  is three times greater than that obtained by using the
is three times greater than that obtained by using the  catalyst.
catalyst.
Article Details

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
-
Attribution — You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
-
ShareAlike — If you remix, transform, or build upon the material, you must distribute your contributions under the same license as the original.
- No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.




